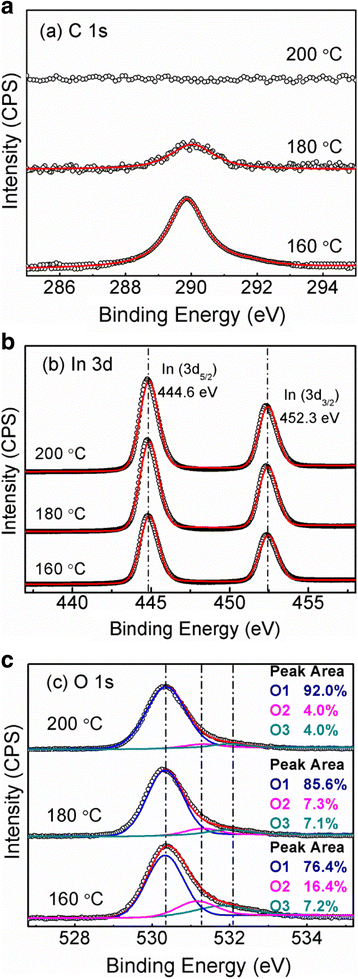
While the large indium metal peaks in the high resolution XPS spectra peak suggest that a significant amount of the indium oxide has been removed, the presence of a large quantity of carbon (indicated in the survey XPS spectrum in FIG. Instrumentation, X-Ray Photoelectron Spectroscopy: An Introduction to Principles and Practices (John Wiley & Sons, Inc. Note the large carbon peak evident at 285 eV. Tauc, Amorphous and Liquid Semiconductors (Springer US Plenum, New York, 1974) (Elsevier Butterworth-Heinemann, Oxford, 2001) Nanostructured materials for biomedical applications (Elsevier, New York, 2017), p. 503 Nanostructured materials for biomedical applications (Elsevier, New York, 2017), p. 411į.
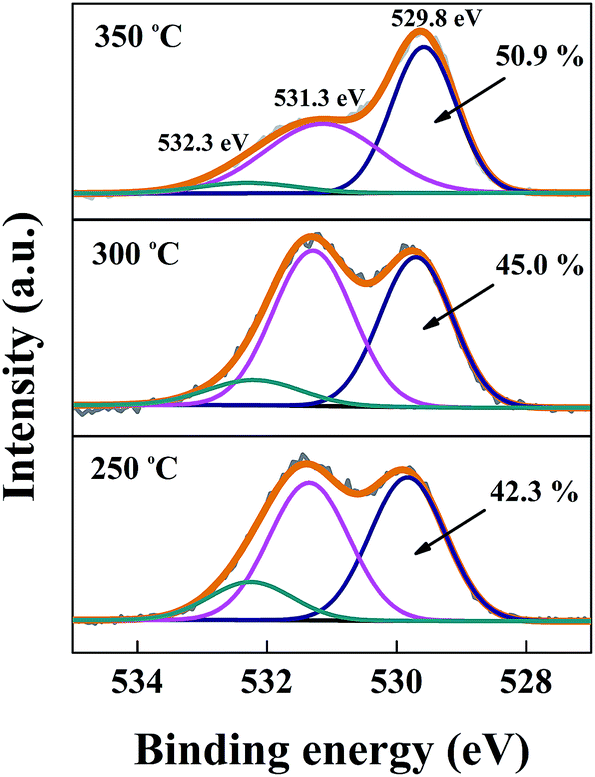
Pérez, Mendoza-Alvarez, in Nanobiomaterials, ed. The effect of the indium myristate precursor concentration on the optical, structural, surface chemical, and electronic properties of InP and QDs will be discussed. To remedy the complication of pressing samples into metal foils, as well as the corresponding spectral contamination, we have successfully used double-stick carbon tape used in SEM sample. From X-ray photoelectron spectroscopy (XPS) analysis, it was confirmed the formation of InP, ZnS, and In 2O 3 moreover, by means of a valence band analysis, the electronic structure of the samples was further investigated. One problem with this method is that indium, being a fairly high-Z material, can provide significant spectral contamination from its many XPS and Auger peaks. From X-ray diffraction (XRD), it was shown the presence of crystalline phases of the InP, ZnS, and In 2O 3 nanoparticles, with sizes ranging from 8 to 10 nm as determined by high resolution transmission electron microscopy (HR-TEM). Furthermore, an enhanced PL emission due to a passivation effect in the ZnS-covered InP QDs was obtained. The Wagner plot shows the binding energy of the XPS In 3d 5/2 peak on the X-axis. The Wagner plot for In, In 2 O 3, In (OH) 3 and In (OH) 3 nH 2 O is shown. From the photoluminescence (PL) spectra, emission peaks located in the range from 2.1 to 3.0 eV were observed. Metallic indium can be differentiated from the indium oxide and hydroxide phases, but the oxide and hydroxide chemical states cant be differentiated using the XPS BE chemical shift alone. The band-gap energy of the samples was determined from the absorption spectra. It was found a color variation of the QDs in solution due to the quantum confinement effects when the nanoparticle sizes are smaller than the exciton Bohr radius.

In this work, we present results on the synthesis and characterization of InP and quantum dots (QDs), grown using a single-step chemical synthesis method without injection of hot precursors, varying the concentration of indium myristate in both cases.
